Intensification of Continuous Ortho-Lithiation at Ambient Conditions—Process Understanding and Assessment of Sustainability Benefits,Organic Process Research & Development - X-MOL

Diastereoselective Synthesis of Axially Chiral Xylose-Derived 1,3-Disubstituted Alkoxyallenes: Scope, Structure, and Mechanism.,The Journal of Organic Chemistry - X-MOL

Synthetic efforts on the road to marine natural products bearing 4- O -2,3,4,6-tetrasubstituted THPs: an update - RSC Advances (RSC Publishing) DOI:10.1039/D0RA10755G

Radical Approach to the Chiral Quaternary Center in Asperaculin A: Synthesis of 9‑Deoxyasperaculin A - презентация онлайн

Synthesis, Reaction, and Recycle of Fluorous Palladium Catalysts for an Asymmetric Allylic Alkylation without Using Fluorous Solvents | The Journal of Organic Chemistry
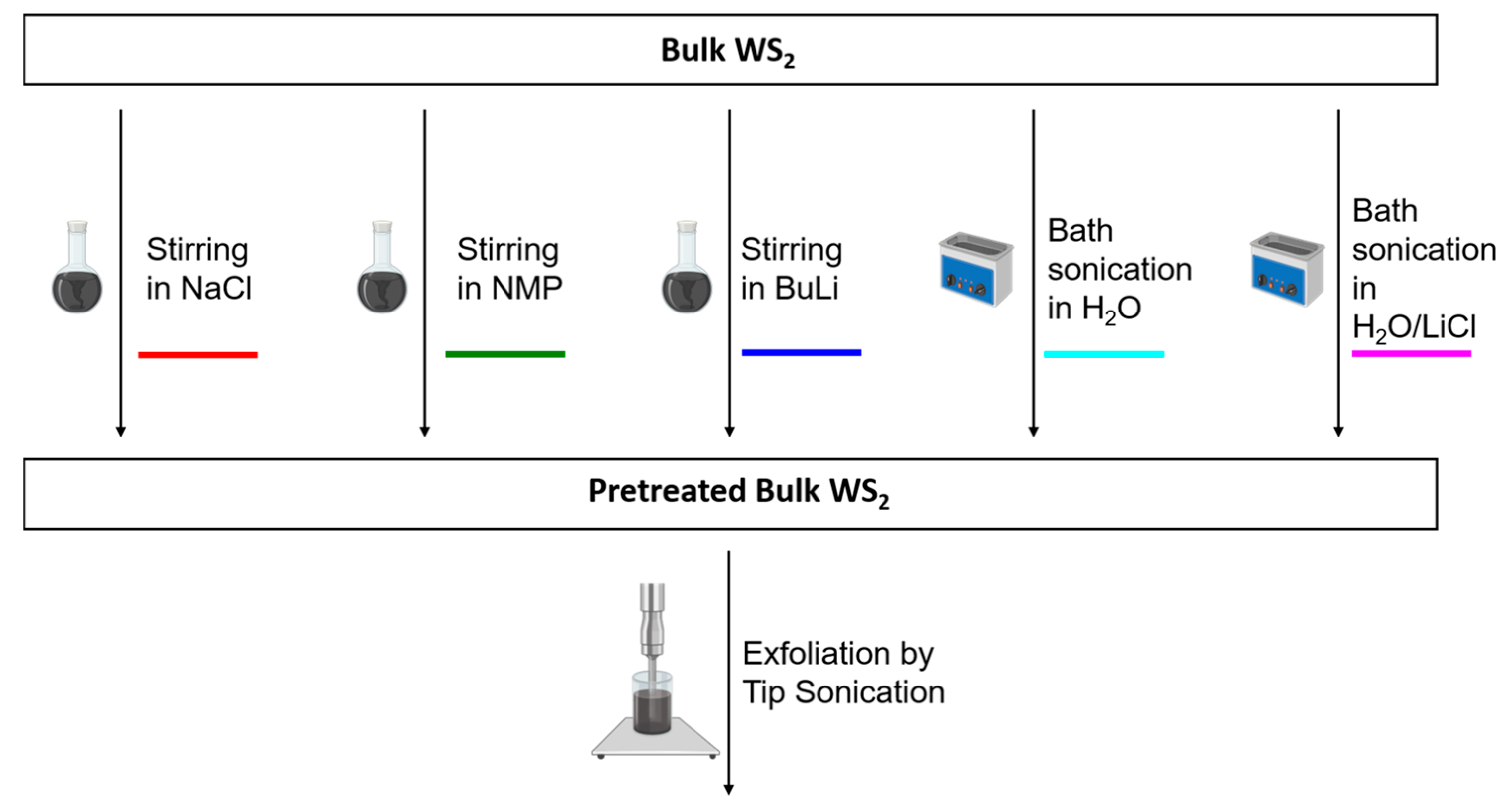
Nanomaterials | Free Full-Text | Impact of Pretreatment of the Bulk Starting Material on the Efficiency of Liquid Phase Exfoliation of WS2 | HTML

Halogen–Lithium Exchange Reaction Using an Integrated Glass Microfluidic Device: An Optimized Synthetic Approach,Organic Process Research & Development - X-MOL
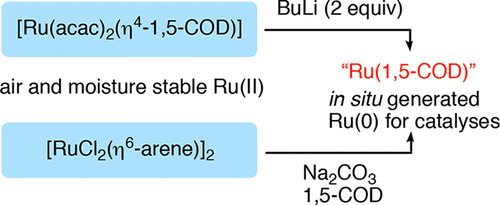
In Situ Routes to Catalytically Active Ru(0) Species by Reduction of Readily Available, Air-Stable Precursors,Organometallics - X-MOL

Cross-Coupling of Alkynylsilanols with Aryl Halides Promoted by Potassium Trimethylsilanolate | The Journal of Organic Chemistry

Generation and Electrophile Trapping of N -Boc-2-lithio-2-azetine: Synthesis of 2-Substituted 2-Azetines – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.
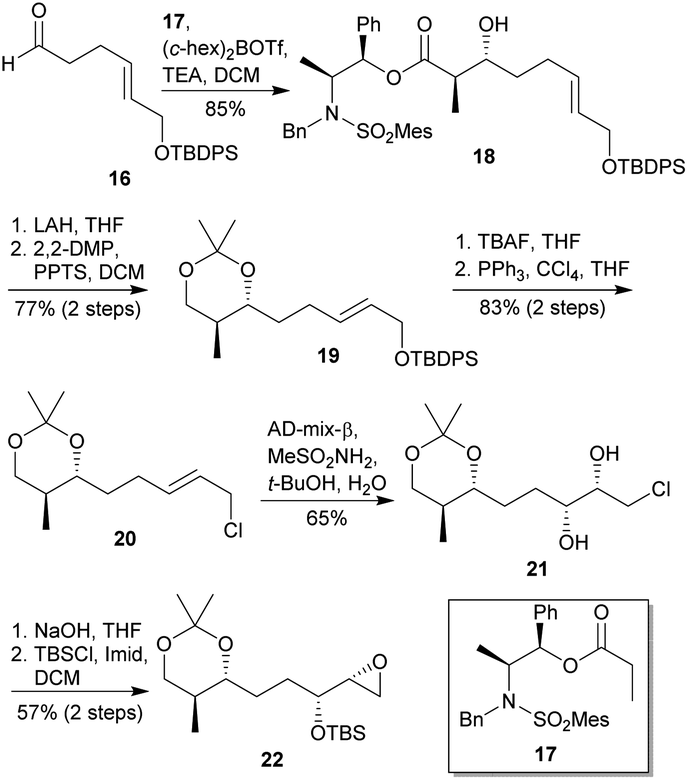
Synthetic efforts on the road to marine natural products bearing 4- O -2,3,4,6-tetrasubstituted THPs: an update - RSC Advances (RSC Publishing) DOI:10.1039/D0RA10755G
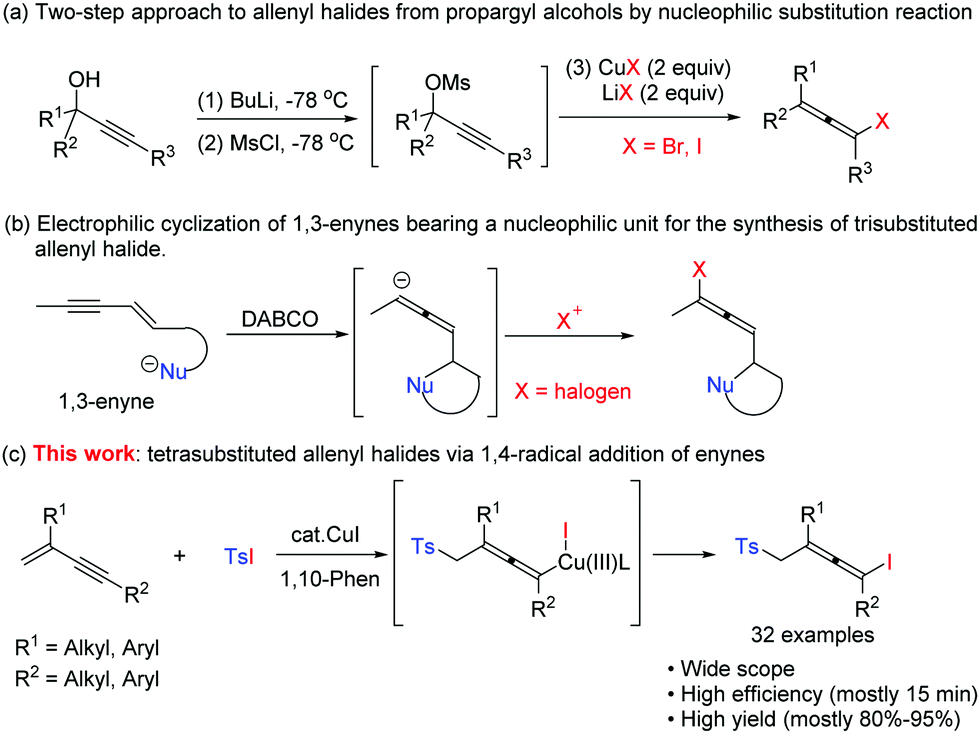
Copper-catalyzed radical approach to allenyl iodides - Chemical Communications (RSC Publishing) DOI:10.1039/C9CC05853B

Report: Exploring the Potential of an Acid-Initiated Vinylogous Aldol Reaction to Form All-Carbon Quaternary Stereocenters (62nd Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)
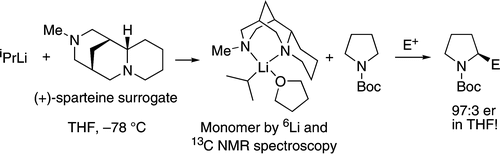
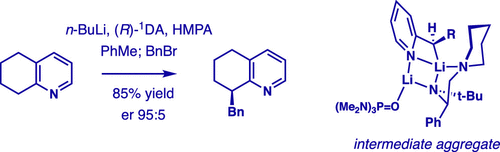
Asymmetric deprotonation using s-BuLi or i-PrLi and chiral diamines in THF: the diamine matters.,Journal of the American Chemical Society - X-MOL
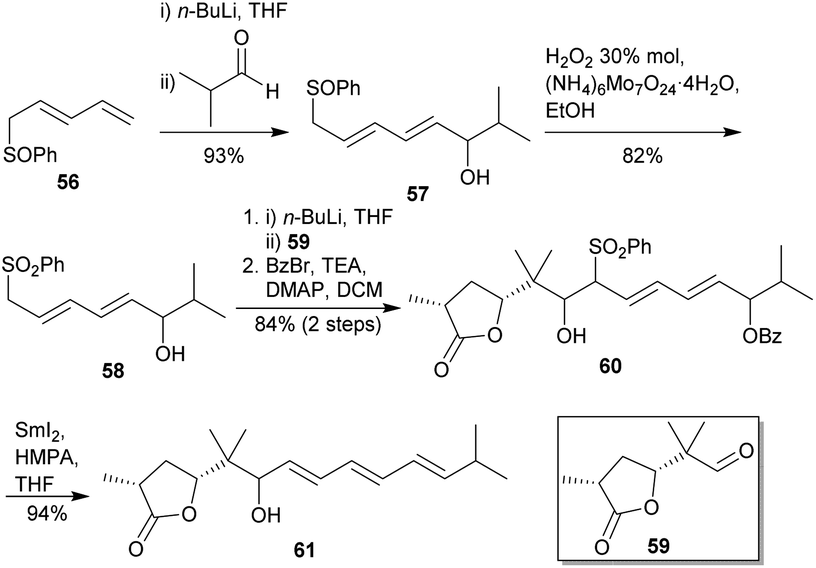
Synthetic efforts on the road to marine natural products bearing 4- O -2,3,4,6-tetrasubstituted THPs: an update - RSC Advances (RSC Publishing) DOI:10.1039/D0RA10755G

PDF) Synthetic Approach to the Core Structure of Oleandrin and Related Cardiac Glycosides with Highly Functionalized Ring D

Polymer Brushes by Living Anionic Surface Initiated Polymerization on Flat Silicon (SiOx) and Gold Surfaces: Homopolymers and Block Copolymers | Langmuir
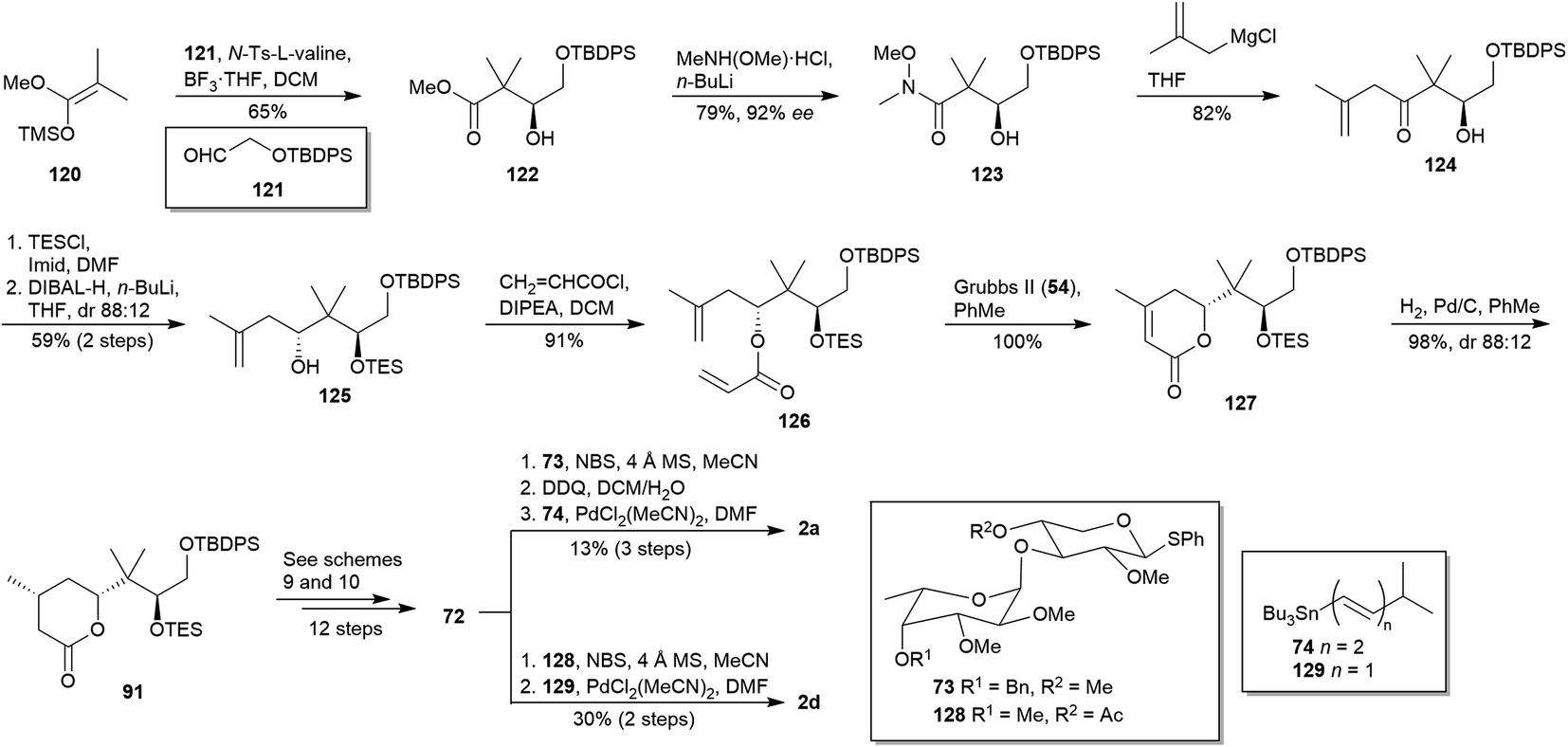
Synthetic efforts on the road to marine natural products bearing 4- O -2,3,4,6-tetrasubstituted THPs: an update - RSC Advances (RSC Publishing) DOI:10.1039/D0RA10755G